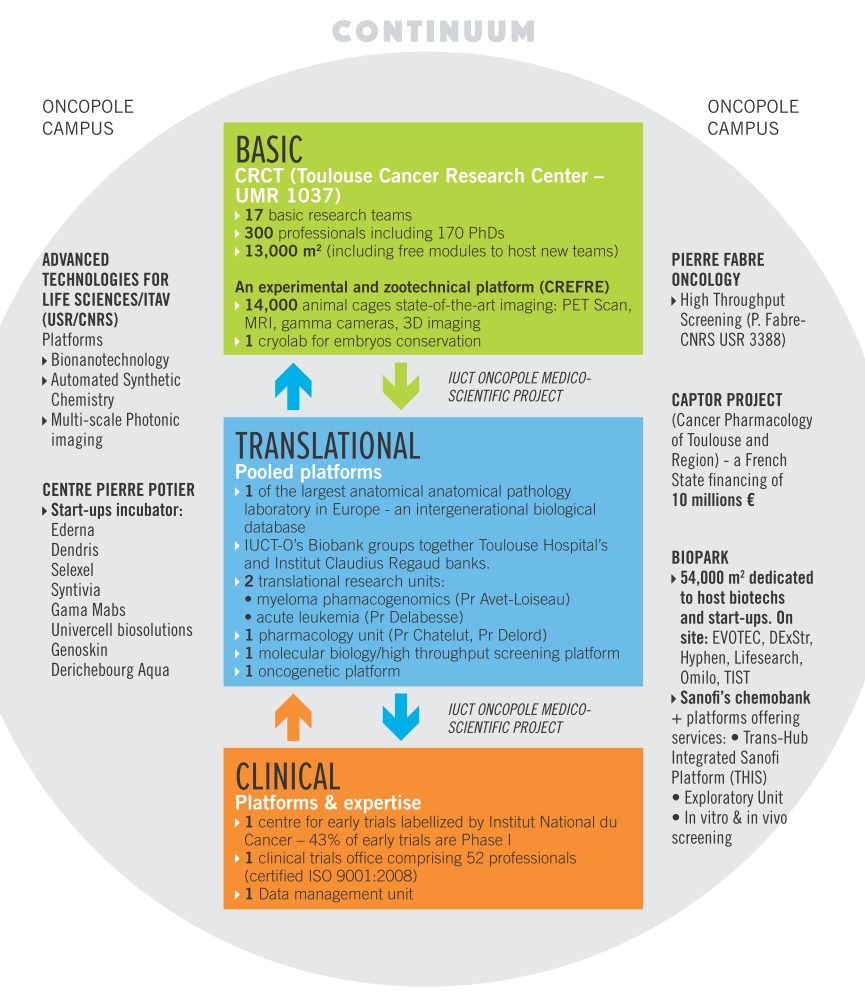
Innovation at the IUCT-O
One of the largest anatomical pathology laboratories in Europe in terms of activity with over 1000 daily analyses, solid and liquid tumors, telepathology (virtual slides), 2 rooms with a decompression chamber for immediate diagnoses of surgical specimens (within 20 minutes).
Pharmacy : 400 chemotherapy operations per day, nuclear pharmacy (routine and research activities)
Hematology : 100 beds and places - 91 clinical trials conducted in 2011
Surgery : 50% of outpatient activities dedicated to senology. An operating room dedicated to 3D surgery (in partnership with Olympus).
Clinical trials : INCa-designated early-phase clinical trial center (CLIP2). The clinical trials administrative center was the first French center to obtain ISO 9001:2008 certification for the design and management of cancer clinical trials in 2012.
A molecular biology platform .
Nuclear imaging : world's first installation of the latest generation PET scan (Discovery IQ - partnership with General Electric Healthcare). Accredited PET/CT scan center of excellence.
60% of radiotherapy accelerators have been renewed. A proton therapy project.
Recherches nécessitant une réutilisation de données
La nouvelle réglementation en vigueur applicable au traitement de données à caractère personnel et, en particulier, le règlement UE 2016/679 du Parlement européen et du Conseil du 27 avril 2016 (ci-après le « RGPD ») a pour objectif de renforcer la maîtrise des personnes sur les données les concernant.
Les personnes prises en charge dans le cadre de l’activité de l’Institut doivent être informées du traitement de données personnelles les concernant (art. 14 RGPD). Les patients sont autorisés à demander à l’Institut la communication des données recueillies et à exercer leurs droits de modification, de correction, de mise à jour ou d’effacement de ces données.
Easier access to innovation
The IUCT-Oncopole gives full priority to innovation and research with the following objectives:
- to promote therapeutic and medical innovation as part of a care continuum, clinical research and basic research so discoveries may be translated into tangible benefits for patients ;
- to help with the transfer of knowledge by establishing links with industrial partners ;
- to disseminate new knowledge and practices amongst health professionals and the general public (in connection with the university).
A structured and multidisciplinary organization for dispensing care
In order for patients to have access to quality care and innovation, the IUCT-O has developed diagnostic and therapeutic technical platforms :
- 2 biological laboratories ;
- grouping together all the anatomical pathology services of Toulouse public sector ;
- the presence of one of the 28 INCa-designated molecular genetics hospital platforms. It allows molecular tests to be performed in order to guide and refine the diagnosis and better target the treatment strategies.
The care-research continuum